The immune system is a defense mechanism that distinguishes from the self and non-self and responds to viruses and bacteria intruding from outside or abnormalities like tumor cells to protect myself. However, the immune system attacks not only the intruder from outside but sometimes normal organisms and cells within the human body. Excessive immune response, manifesting from breaking the balance of the immune system leads to severe diseases, and it’s a major factor that can diminish the quality of life.
Autoimmune disease is a condition in which imbalance on the immune system causes an excessive immune response to the autoantigens, secrete an excessive number of inflammatory signals to cause abnormalities in immune responses. Autoimmune disease may emerge acutely or chronically and can affect all organs and organisms, exhibiting systemic symptoms including systemic malaise, fatigue, mild fever, changes in weight, dry eyes or mouth, ulcers, joint pain, rashes on the skin. The origin of autoimmune diseases is yet to be clearly identified, while recent studies suggest that autoimmune diseases are related to T cells, a type of lymphocytes among white blood cells.

Especially, about 80% of autoimmune diseases manifest in women, and the prevalence in young women or children are rising recently, as well as the number of male patients. Other than the well-known Rheumatoid Arthritis, Multiple Sclerosis, Lupus, Type I diabetes and thyroiditis, about 100 kinds of autoimmune diseases have been identified. However, there is no fundamental treatment other than administering steroids to control inflammation and immuno-suppressive drug to suppress the excessive immune response, and the development of a remedy to improve the survival rate and quality of life of the patients is imperative.

Since Novatis’ Kymriah (Chimeric Antigen Receptor-T, CAR-T) was approved by FDA as cellular immunotherapy for acute lymphoblastic leukemia, commercialization of cellular immunotherapy has been increasing, and the Korean market for immune cellular medicines are growing. Especially, after the Act on safety and support of cutting-edge restorative medicine and biopharmaceuticals came into law in September 2020 Kymriah (CAR-T) by Novatis was the first medication that was approved by the Korean Ministry of Food and Drug Safety in May 2021. Curocell also recently started clinical trials for a CAR-T.
Cellular immunotherapy is a medicine produced through chemical, biological manipulation, exterior cultivation or reproduction of immune cells obtained from blood or cord blood. Unlike the existing stem cell medications, Cellular immunotherapy can be used primarily for the treatment of cancer or autoimmune diseases. Especially, if health immune cells can be commercialized as needed in off-the-shelf form through developing allogeneic cellular immunotherapy, it is expected that the cost of production, as well as administration, will decrease.
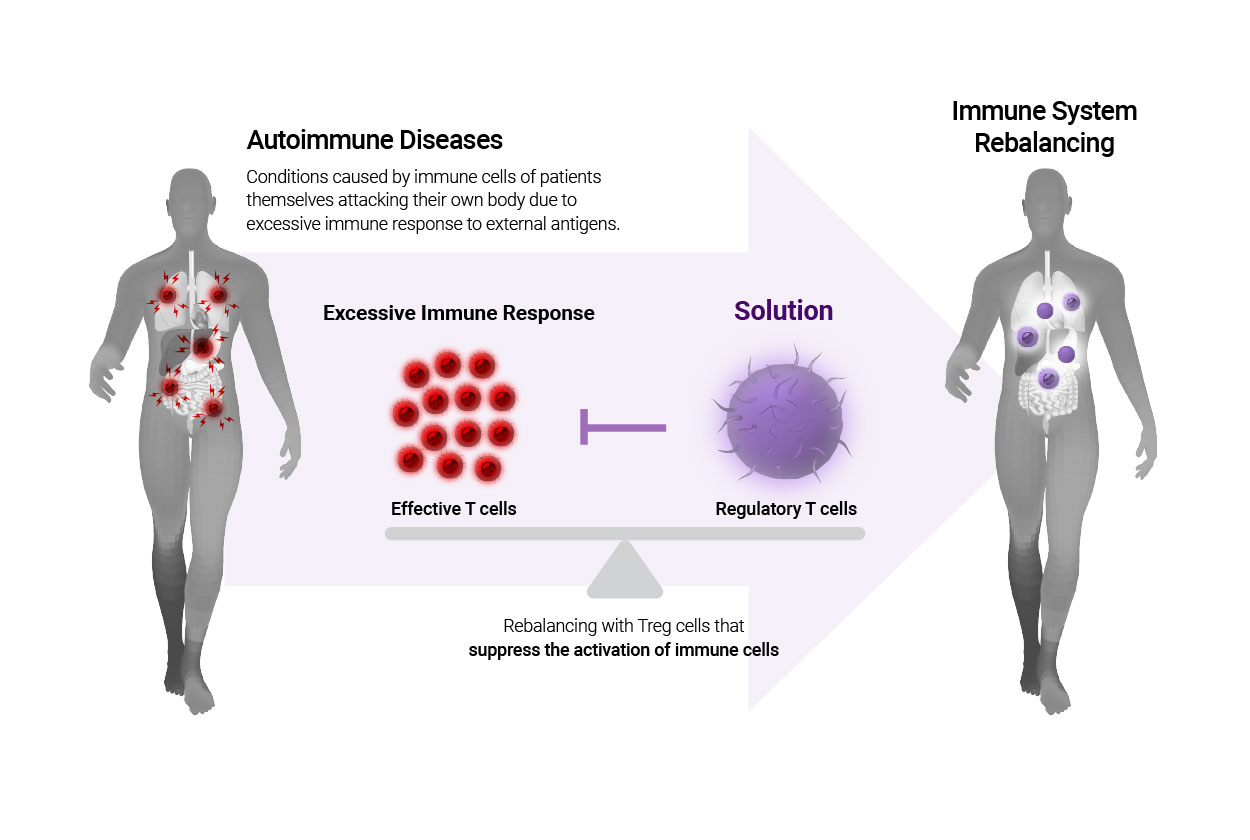
Regulatory T cells (Treg cells) is a type of immune cell that maintains immune tolerance on autoantigens, and they suppress the activation of cytotoxic T cells which attack antigens. Many companies in Korea and around the globe are attempting to develop a remedy for autoimmune diseases using regulatory T cells. The market for autoimmune disease medications is expected to grow up to USD 1.5 trillion in 2025, and the market for cellular medication is estimated to grow to USD 48.1 billion by 2027. In the market for cellular immunotherapies, Treg cellular therapy appear the last, trailing T cells and NK cells; however, they retain high efficacy in treating autoimmune diseases and became a crucial field of research in cellular gene therapy market.
Recently, a specialized global bio enterprise that develops Treg cellular therapy recently sees a drastic increase. Numerous companies, including Sangamo, TRexBio, Kyverna, Sonoma, Quell, ActiTrexx, GentiBio, Abata, Teraimmune, Cellenkos in the United States, GC Greencross Cell, GI Cell, Immunis Bio in Korea are challenging to the development of medication for autoimmune disease using Treg cellular therapy. Especially, these companies aim to overcome autoimmune diseases by establishing a Treg cellular treatment regimen with the introduction of genetic editing technology after the manifestation of CAR or TCR upon target organic antigens.
Using Treg cells isolated from cord blood, IMMUNIQUE is challenging to overcome not only hyperimmune diseases such as Graft versus Host Disease and aplastic anemia but also other autoimmune diseases through developing antigen-specific Treg therapy and CAR-Treg cellular therapy. The company sets itself apart from competing global bio companies through immune cellular remedies derived from allogeneic samples using a stem cell platform and is galloping towards the research towards overcoming autoimmune diseases.