Traditional methods to treat patients with tumors include surgery, chemotherapy and radiotherapy. However, in many cases, these methods fail to cure tumors completely due to their inherent limitation. Even if tumor tissues are removed through surgery, there is a high risk that a small number of tumor cells cause metastasis onto other parts of the body. While chemotherapy could be utilized throughout a relatively wider range, the rate of cure is not good in most solid tumors, in addition to the side effect as the medicine tends to kill other ordinary cells.
The development and commercialization of immunochemotherapy are seeing a rapid increase to overcome the limitations of these conventional oncological treatment strategies. Immunochemotherapy stimulates the immune system so that immune cells selectively attack tumor cells. Types of medications include immune checkpoint inhibitor, which revitalizes the immune cells, and antigen therapy and Cellular Immunotherapy that targets tumor antigens to attack.

Anticancer cellular immunotherapy is a medication that treats intractable diseases such as tumors, by injecting immune cells of the human body after cultivating, amplifying and reinforcing outside the human body. This method activates the immune function of the human body, and the advantage is that the patients eliminate tumor cells within their body using their own immune system and prevent relapses. Moreover, it sets itself apart from existing treatment strategies as it is highly customized to an individual patient.
Anticancer cellular immunotherapy underwent active research centred around the US including an attempt of clinical research by the National Institute of Health on malignant melanoma patients using autologous T-cells activated through exterior cultivation in the 80s. However, T-cells without antigen specificity cannot completely remove malignant melanoma. To provide antigen specificity to T-cell, Chimeric antigen receptor has been employed combining the antigen recognition part (scFv) and T-cell activation domain within cells.
Especially, CD19-CAR-T cells shortened the timeframe of commercialization of cellular immunotherapy as it performed well in a clinical trial for relapse cases of pediatric patients suffering from malignant B-cell leukemia. Despite these results, the property of T-cells that undergo rapid clonal expansion upon encountering target cells tend to cause cytokine storm, and if T-cells are injected into another person they act as an immunogen. Therefore, it is a barrier for commercialization because the method is essentially extravagant therapy designed for just one person costing up to hundreds of millions of Korean Won.
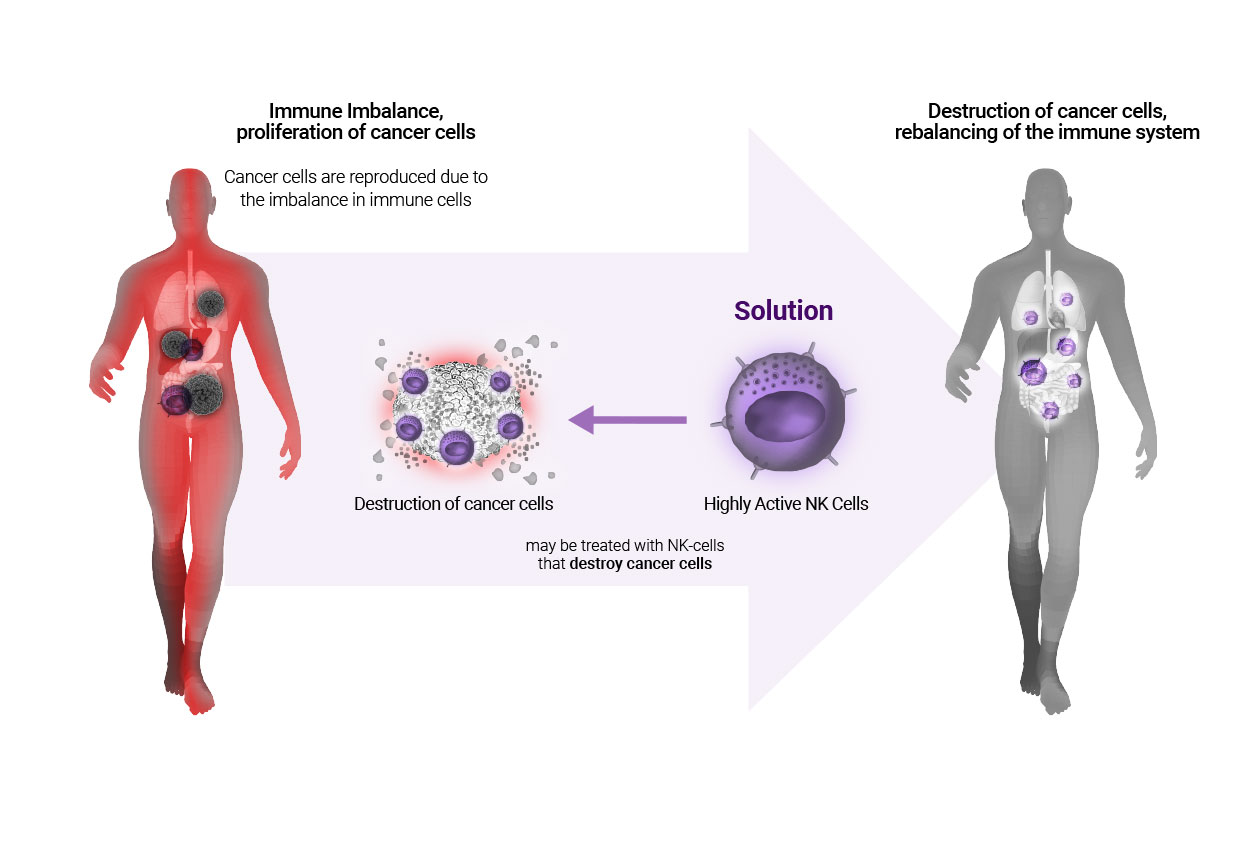
The commercialization of CAR-T cells impacted the development of Natural Killer Cell therapy. NK cells are immunocytes that immediately recognize and eliminate cells infected with virus or tumor cells, and they are the effective cells that act as the first line of defense for our body. Compared to T-cells, clonal expansion or memory function is weaker whereas they pose less risk for Cytokine storm. In transplantation of hematopoietic stem cells in cases of leukemia, injection of NK cells suppressed graft-versus-host disease (GvHD) and induced Graft-versus Leukemia, GvL). Therefore, clinical trials using NK cells which had been limited to leukemia treatments, are now being performed on melanoma, lymphoma, and solid cancers in Korea and around the world.
The size of the cellular therapy market across the globe including NK cells drastically jumped from USD 4 billion in 2015 to USD 7.5 billion in early 2020, and the market based on NK cells is projected to grow from about USD 1.5 billion in 2018 to 3.8 billion in 2026. The development of NK cellular therapy in the US is performed by various bio enterprises including Fate Therapeutics, Nkarta Inc, Affimed NV, ImmunityBio, Anthrogenesis, Artiva Biotherapeutics, and others. Moreover, many players are actively engaging in the development, such as GC Green Cross Lab Cell which is developing therapies targeting 3 types of solid cancer in association with Merck & Co., NKmax which possesses original technology platform ‘Super NK,’ capable of mass cultivation of NK cells derived from high purity peripheral blood, BigCell Bio, which utilizes NK cells, congenital immune cells, instead of genetic manipulation technologies, SMT Bio which performs combined treatment with NK cells with Pembrolizumab, Cha Biotech, which improved reproduction capacity of NK cells by 2000 folds, and Genexin which is currently in co-development project with Toolgen for ‘Crispr’ allogenic ‘CAR-NK.’
IMMUNIQUE is developing anticancer immunotherapy targeting not only leukemia but also solid cancers through mass reproduction of NK cells derived from cord blood. Moreover, the company is also focusing on a development project aiming to overcome the ageing of NK cells using proteins and introduce genes to attack B-cell Lymphoma.